
Too many aggressive prostate cancers are missed
Prostate cancer is a major cause of cancer-related deaths. Current screening with PSA misses aggressive cancers in men with low PSA levels. Higher PSA-levels are often unrelated to cancer.
• Designed for early detection and screening
• 60% fewer prostate cancer deaths at six years follow-up
• Identifies aggressive cancer missed by PSA
• 90% sensitivity for aggressive disease
• Designed for early detection and screening • 60% fewer prostate cancer deaths at six years follow-up • Identifies aggressive cancer missed by PSA • 90% sensitivity for aggressive disease
The limitations of PSA screening
PSA screening has saved lives by detecting aggressive cancers early, but it also has important limitations.
A major problem is that it misses aggressive cancers in men with low PSA levels (below 3-4 ng/mL). Up to 50% of aggressive prostate cancers occur at low PSA levels.
PSA can also rise for reasons unrelated to cancer, and has limited ability to distinguish between slow-growing, harmless cancers and aggressive cancers that require treatment. This leads to false alarms causing unnecessary magnetic resonance imaging (MRI) scans, biopsies, anxiety, and healthcare costs.


Up to 50% of aggressive cancers occur at low PSA levels (<3 ng/ml)
A next generation screening test
Stockholm3 is a blood-based test that combines protein biomarkers, genetic risk, and clinical factors to estimate a man’s risk of having aggressive prostate cancer.
Stockholm3 can replace PSA as a more precise screening tool by finding aggressive cancers (defined as ISUP Grade Group ≥ 2) earlier while reducing the over-detection that has historically limited prostate cancer screening.
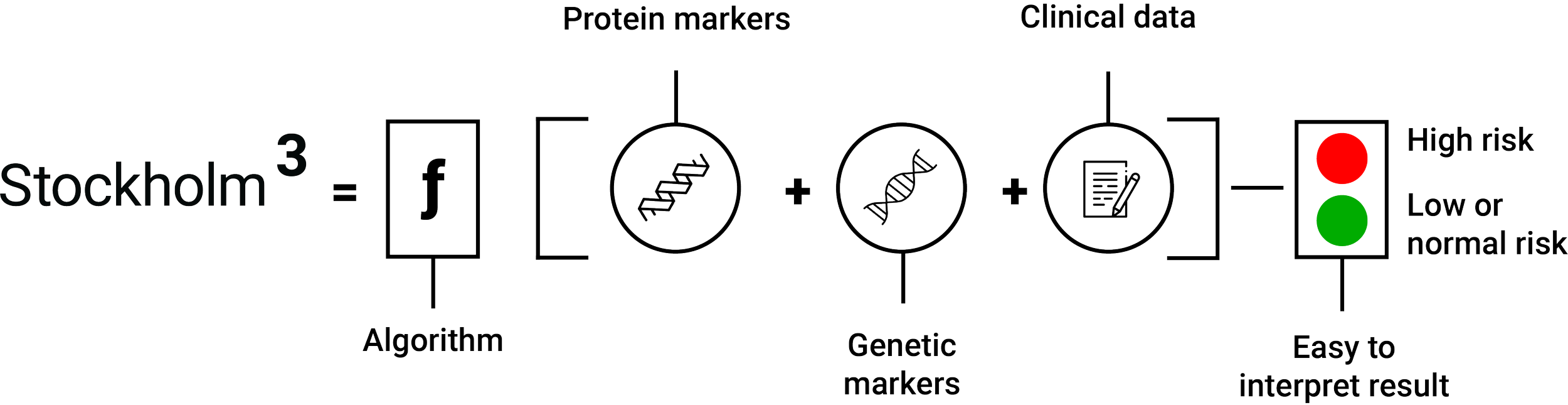
By clarifying risk earlier in the screening process, Stockholm3 enables more appropriate follow-up. Men at higher risk can be identified earlier, while men at low risk can avoid unnecessary diagnostic pathways.
Finding More Aggressive Cancers When They Are Curable
Improved screening of aggressive cancer
Identifies aggressive disease missed by standard PSA thresholds
Fewer unnecessary investigations in low-risk men
Stage migration towards more curable disease
Built for Large-Scale Clinical Implementation
Stockholm3 uses a standard blood draw and integrates into existing clinical workflows. It provides an individualized Stockholm3 Risk Score.
An elevated Stockholm3 Risk Score indicates an increased risk for aggressive prostate cancer and may support further diagnostic workup, such as MRI or prostate biopsy, in accordance with current medical practice.

Validated by Large-Scale Clinical Evidence
Stockholm3 has been evaluated in high quality studies with more than 90,000 men and in clinical routine care.
Findings from prospective screening studies, large-scale real-world implementation and long-term follow-ups point in the same direction: improved early detection of aggressive prostate cancer, better risk discrimination, and fewer prostate cancer related deaths.



Improved Screening of Aggressive Prostate Cancer
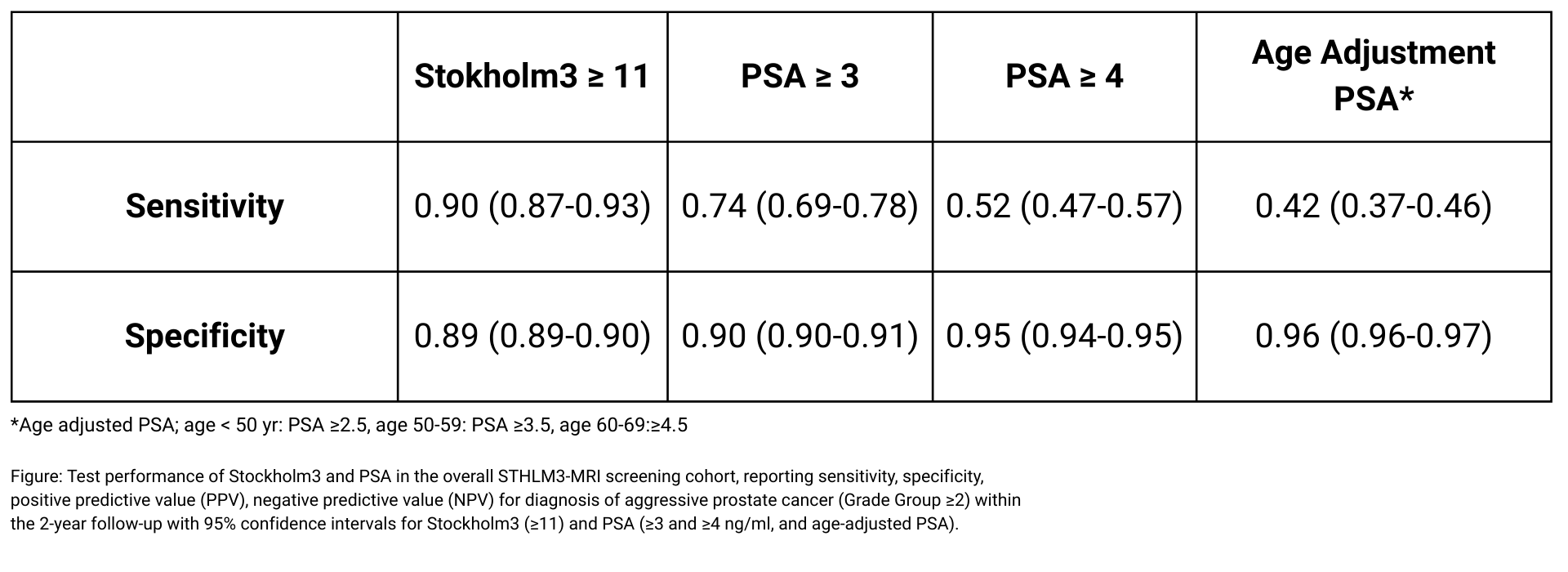
In a large prospective screening study (N=12,750), Stockholm3 demonstrated 90% sensitivity for detecting aggressive prostate cancer. This performance significantly outperforms PSA thresholds at comparable specificity.
Follow-up from screening studies suggests evidence of 60% reduced prostate cancer mortality when Stockholm3 is used compared with PSA-based screening.
This improvement in survival appears to be driven by earlier detection of biologically aggressive cancers that are frequently missed by conventional PSA use.
Long term follow-up suggests reduced prostate cancer deaths
Real-World Performance at Scale
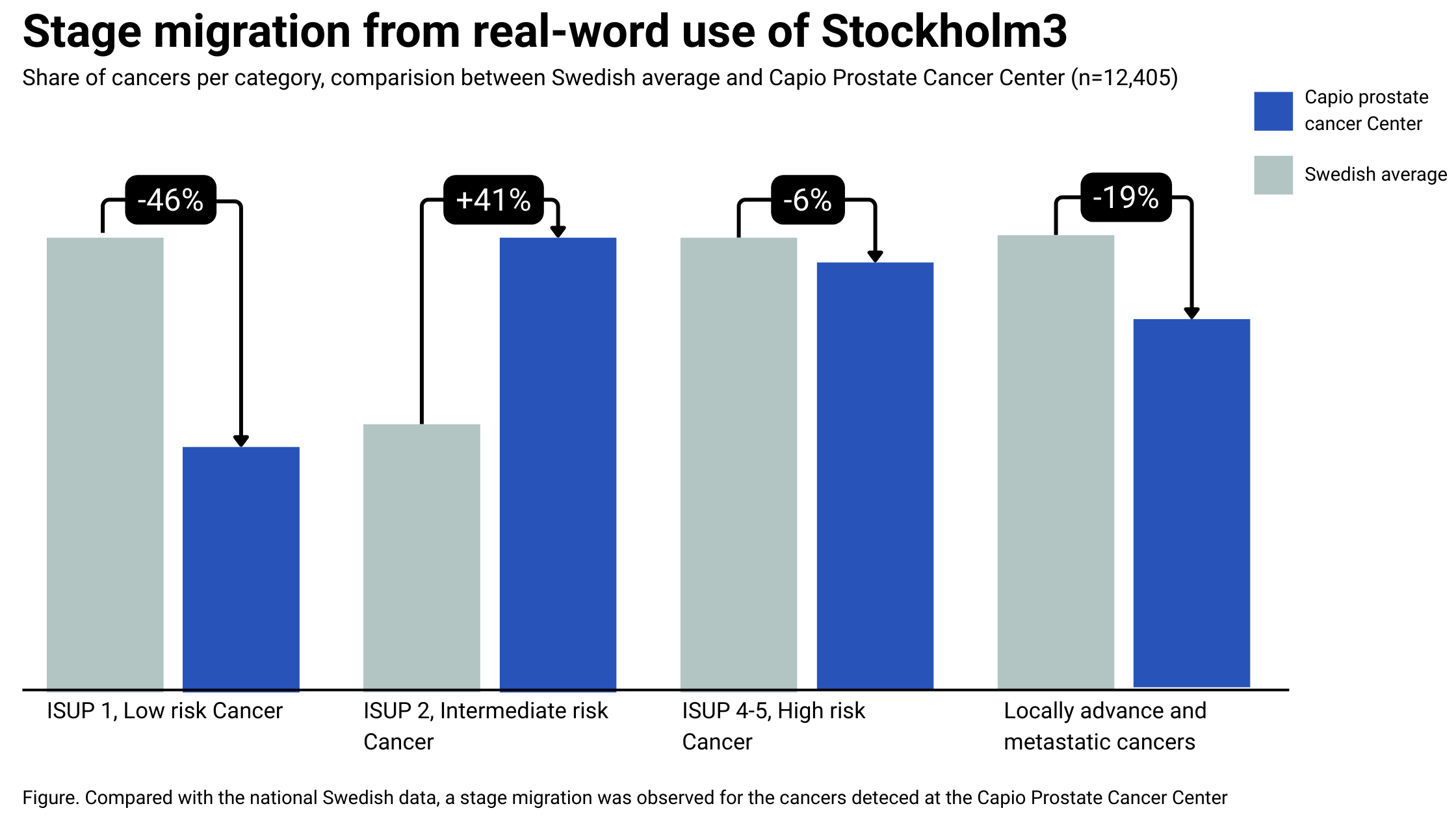
Real-world use of Stockholm3 has led to stage migration toward more curable disease and less indolent and advanced disease.

Real-World use Norwegian health system (N=4,784)
Replacing PSA with Stockholm3 led to an 89% increase in the detection of aggressive prostate cancer and a 26% reduction in the detection of indolent disease.
Real-world use Swedish Health system (N=12,405)
Replacing PSA with Stockholm3 led to stage migration toward more curable disease and less indolent and advanced disease.
Validated Across Diverse Populations
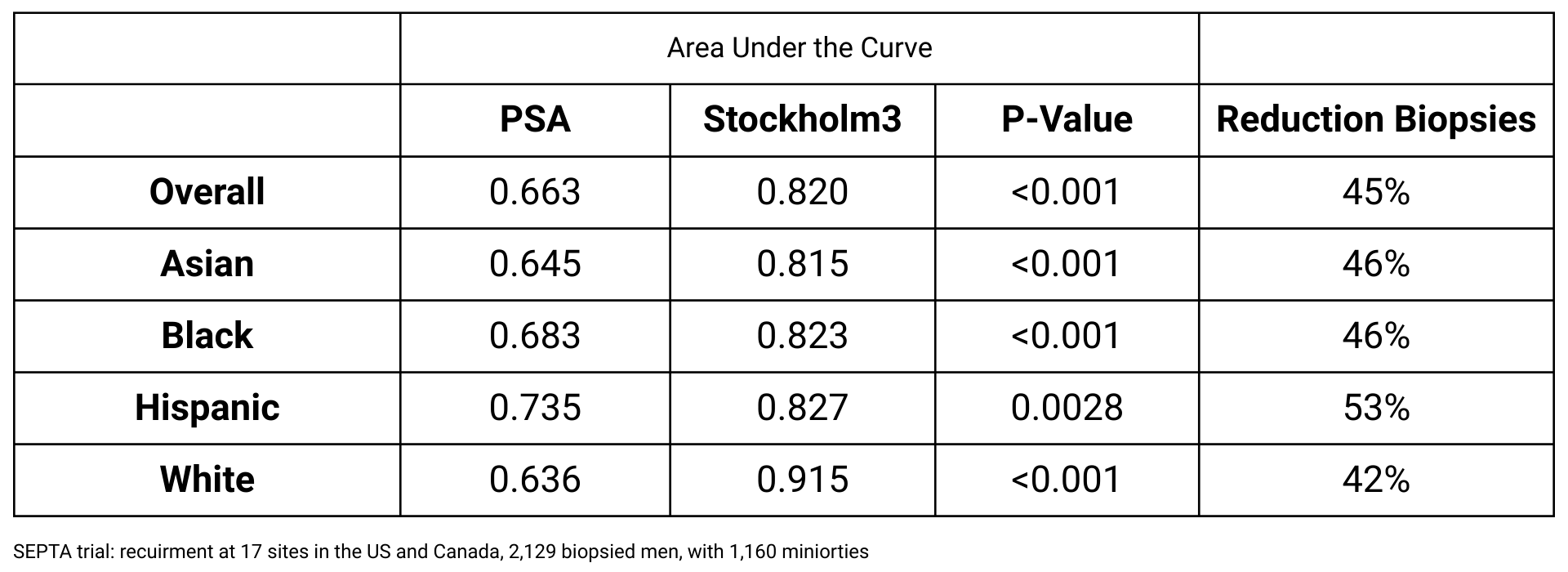
Stockholm3 has been prospectively validated in multiethnic cohorts in the U.S. and Canada, representing the largest evaluation of a prostate cancer biomarker in minority populations.
The test demonstrates equivalent diagnostic accuracy across all racial and ethnic groups, ensuring consistent risk discrimination for broad population screening.
FAQs
-
Stockholm3 is a blood-based test, that runs a combination of protein biomarkers, genetic biomarkers and clinical information through an algorithm to find the probability of clinically significant cancer at biopsy.
Stockholm3 has been evaluated in clinical studies including more than 90,000 men. It is extensively tested in large population-based screening trials, as well as in real world clinical utility studies in primary care, as a reflex test to PSA at values 1.5-20 ng/ml. Benefits include the ability to reduce unnecessary MRIs, benign and Grade Group 1 prostate biopsies for men with elevated PSA, while simultaneously improving detection of clinically significant cancers in men with low or normal PSA values. Using Stockholm3 leads to a more accurate risk assessment than the current PSA standard. Stockholm3 detects 41-89% more men with aggressive prostate cancer and, at the same time, reduces over-detection by 37-52% compared to PSA.
Multiple Stockholm3 studies have been published in high-impact journals such as The Lancet Oncology, Journal of Clinical Oncology, and European Urology. The studies address both the specificity and sensitivity of Stockholm3 in multi-ethnic populations as well as health-economic benefits of implementing it in clinical care. [2, 3]
-
Stockholm3 was developed by researchers at the Karolinska Institute in Stockholm, Europe’s leading medical university. It has been evaluated in clinical studies with more than 90,000 men of different ethnicities, including studies in the United States, Canada, Germany, Sweden, Norway, and Switzerland.
-
Your test result will indicate whether you have a low, normal, or elevated risk of prostate cancer. If you have a normal or low risk, you will be recommended another prostate cancer test in two or six years, respectively (Europe only). If you have an elevated risk, you will be recommended further examination with a magnetic camera and/or a biopsy in consultation with the attending physician.
-
Stockholm3 is currently available in the following markets: The UK, Switzerland, Germany and the Nordic countries. For information on how to order the test, please contact:
Europe
Germany: Labor Dr. Wisplinghoff, e-mail: labor@wisplinghoff.de; telephone: +49 (0) 221 940 5050
Switzerland: labor team, e-mail: info@team-w.ch; telephone: +41 71 844 45 45
UK: The Doctors Laboratory, e-mail: stockholm3@tdlpathology.comTelephone +44(0) 207 307 7373
Nordics (Sweden, Norway, Finland): A3P Lab Customer Service, Uppsala, Sweden. E-mail: customersupport@a3p.com, telephone: +46 (0) 10-182 33 01
All other markets
A3P Lab customer service, Uppsala, Sweden.
E-mail: customersupport@a3p.com, telephone: +46 (0) 10-182 33 01
-
1. Grönberg et al. Prostate cancer screening in men aged 50–69 years (STHLM3): a prospective population-based diagnostic study. The Lancet Oncology. 2015
Nordström et al. Prostate Cancer Screening Using a Combination of Risk-Prediction, MRI, and Targeted Prostate Biopsies. The Lancet Oncology. 2021
Grönberg et al. Prostate Cancer Diagnostics Using a Combination of the Stockholm3 Blood Test and Multiparametric Magnetic Resonance Imaging. European Urology. 2018
Viste et al. Effects of replacing PSA with Stockholm3 for diagnosis of clinically significant prostate cancer in a healthcare system - the Stavanger experience. SJPHC. 2020
Palsdottir, T et. al. The Capio Prostate Cancer Center Model for Prostate Cancer Diagnostics – Real world evidence from 2018 to 2022. European Urology Opean Science. 2024 Jan 25
Elyan et al. Prospective Multicenter Validation of the Stockholm3 Test in a Central European Cohort. Eur Urol Focus. 2023 Oct 7
Fredsoe et al. Results from the PRIMA Trial: Comparison of the STHLM3 Test and Prostate-specific Antigen in General Practice for Detection of Prostate Cancer in a Biopsy-naïve Population. Eur Urol Oncol. 2023 Oct
Vigneswaran et al. Stockholm3 in a Multiethnic Cohort for Prostate Cancer Detection (SEPTA): A Prospective Multicentered Trial. J Clin Oncol. 2024 Jul 22
Tilki et al. External Validation of Stockholm3 in a Retrospective German Clinical Cohort. Eur Urol Focus. 2024 Aug 6
2. Viste et al. Effects of replacing PSA with Stockholm3 for diagnosis of clinically significant prostate cancer in a healthcare system - the Stavanger experience. SJPHC. 2020
Palsdottir, T et. al. The Capio Prostate Cancer Center Model for Prostate Cancer Diagnostics – Real world evidence from 2018 to 2022. European Urology Opean Science. 2024 Jan 25
3. Grönberg et al. Prostate cancer screening in men aged 50–69 years (STHLM3): a prospective population-based diagnostic study. The Lancet Oncology. 2015Eklund et al. The Stockholm-3 (STHLM3) Model can Improve Prostate Cancer Diagnostics in Men Aged 50–69 yr Compared with Current Prostate Cancer Testing. European Urology Focus. 2016
Grönberg et al. Prostate Cancer Diagnostics Using a Combination of the Stockholm3 Blood Test and Multiparametric Magnetic Resonance Imaging. European Urology. 2018
Waldén M et al. A Head-to-head Comparison of Prostate Cancer Diagnostic Strategies Using the Stockholm3 Test, Magnetic Resonance Imaging, and Swedish National Guidelines: Results from a Prospective Population-based Screening Study. European Urology Open Science. 2022
Vigneswaran et al. Stockholm3 validation in a multi-ethnic cohort for prostate cancer (SEPTA) detection: A multicentered, prospective trial. JCO supplements, ASCO-GU 2024 presentation, 2024, Jan 25. Vigneswaran et al. Stockholm3 in a Multiethnic Cohort for Prostate Cancer Detection (SEPTA): A Prospective Multicentered Trial. J Clin Oncol. 2024 Jul 22
Tilki et al. External Validation of Stockholm3 in a Retrospective German Clinical Cohort. Eur Urol Focus. 2024 Aug 6
4. Bergman et al. Structured care for men who want to get tested for prostate cancer – findings from Capio Prostate Cancer Center. Läkartidningen. 2018;115:FCDTViste et al. Effects of replacing PSA with Stockholm3 for diagnosis of clinically significant prostate cancer in a healthcare system - the Stavanger experience. SJPHC. 2020, Söderbäck et al. Improved prostate cancer diagnostics with a structured pathway including Stockholm 3 test, MRI and targeted perineal biopsies. Läkartidningen. 2023
Mcleod et al. Cost Effectiveness of Prostate Cancer Testing incorporating Stockholm3 and MRI versus standard of care. EAU 2024, April 7
Hao S et al. Cost-effectiveness of Stockholm3 test and magnetic resonance imaging in prostate cancer screening: a microsimulation study. European Urology. 2022.
5. Thompson et al. Prevalence of Prostate Cancer among Men with a Prostate-Specific Antigen Level ≤4.0 ng per Milliliter. NEJM 2004
Naji et al. Digital Rectal Examination for Prostate Cancer Screening in Primary Care: A Systematic Review and Meta-Analysis. Ann Fam Med, 2018
Krilaviciute et al. Digital Rectal Examination Is Not a Useful Screening Test for Prostate Cancer. European Urology Oncology, 2023
6. Eggener S. Comment - Prostate cancer screening in men aged 50 to 69 years (STHLM3): A prospective population-based diagnostic study. Urol Oncol. 2017 Mar
7. Grönberg et al. Prostate cancer screening in men aged 50–69 years (STHLM3): a prospective population-based diagnostic study. The Lancet Oncology. 2015
Nordström et al. Prostate Cancer Screening Using a Combination of Risk-Prediction, MRI, and Targeted Prostate Biopsies. The Lancet Oncology. 2021
Grönberg et al. Prostate Cancer Diagnostics Using a Combination of the Stockholm3 Blood Test and Multiparametric Magnetic Resonance Imaging. European Urology. 2018
Viste et al. Effects of replacing PSA with Stockholm3 for diagnosis of clinically significant prostate cancer in a healthcare system - the Stavanger experience. SJPHC. 2020
Palsdottir, T et. al. The Capio Prostate Cancer Center Model for Prostate Cancer Diagnostics – Real world evidence from 2018 to 2022. European Urology Opean Science. 2024 Jan 25
Elyan et al. Prospective Multicenter Validation of the Stockholm3 Test in a Central European Cohort. Eur Urol Focus. 2023 Oct 7
Fredsoe et al. Results from the PRIMA Trial: Comparison of the STHLM3 Test and Prostate-specific Antigen in General Practice for Detection of Prostate Cancer in a Biopsy-naïve Population. Eur Urol Oncol. 2023 Oct
Vigneswaran et al. Stockholm3 in a Multiethnic Cohort for Prostate Cancer Detection (SEPTA): A Prospective Multicentered Trial. J Clin Oncol. 2024 Jul 22
Tilki et al. External Validation of Stockholm3 in a Retrospective German Clinical Cohort. Eur Urol Focus. 2024 Aug 6
Vigneswaran et al. Stockholm3 validation in a multi-ethnic cohort for prostate cancer (SEPTA) detection: A multicentered, prospective trial. JCO supplements, ASCO-GU 2024 presentation, 2024, Jan 25
Waldén M et al. A Head-to-head Comparison of Prostate Cancer Diagnostic Strategies Using the Stockholm3 Test, Magnetic Resonance Imaging, and Swedish National Guidelines: Results from a Prospective Population-based Screening Study. European Urology Open Science. 2022
Söderbäck et al. Improved prostate cancer diagnostics with a structured pathway including Stockholm 3 test, MRI and targeted perineal biopsies. Läkartidningen. 2023,
Mcleod et al. Cost Effectiveness of Prostate Cancer Testing incorporating Stockholm3 and MRI versus standard of care. EAU 2024, April 7
Hao S et al. Cost-effectiveness of Stockholm3 test and magnetic resonance imaging in prostate cancer screening: a microsimulation study. European Urology. 2022
Mottet et al. EAU-EANM-ESTRO-ESUR-SIOG Guidelines on Prostate Cancer-2020 Update. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur Urol. 2021
Wei, J.T., et al., Early Detection of Prostate Cancer: AUA/SUO Guideline Part I: Prostate Cancer Screening. J Urol, 2023.
8. Thompson et al. Prevalence of Prostate Cancer among Men with a Prostate-Specific Antigen Level ≤4.0 ng per Milliliter. NEJM 2004
Hamdy et al. Fifteen-Year Outcomes after Monitoring, Surgery, or Radiotherapy for Prostate Cancer. NEJM, 2023
Loeb, et al. Systematic review of complications of prostate biopsy. Eur Urol. 2013
9. Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023 Jan;73(1):17-48. doi: 10.3322/caac.21763. PMID: 36633525, Cancer Facts and Figures, American Cancer Society, 2023
10. Viste et al. Effects of replacing PSA with Stockholm3 for diagnosis of clinically significant prostate cancer in a healthcare system - the Stavanger experience. SJPHC. 2020
Palsdottir, T et. al. The Capio Prostate Cancer Center Model for Prostate Cancer Diagnostics – Real world evidence from 2018 to 2022. European Urology Opean Science. 2024 Jan 25
